  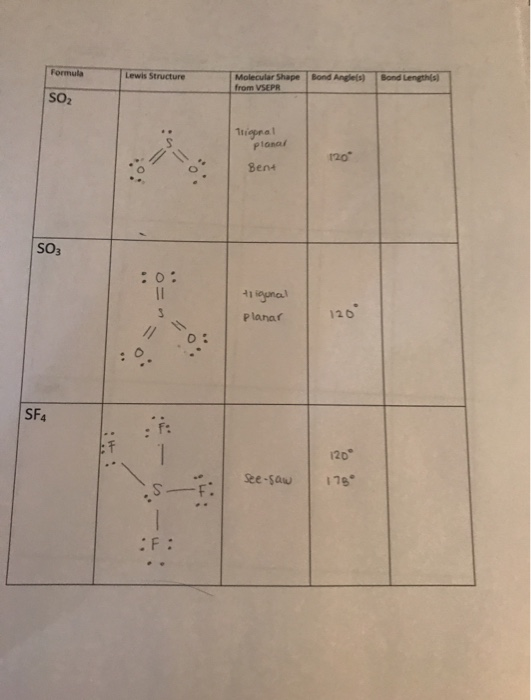
What is the molecular structure of SnCl − 3? XeF4 is a nonpolar molecule and has sp3d2 hybridization. The molecule has octahedral electron geometry and square planar molecular geometry. It has two lone pairs of nonbonding electrons on the central atom of Xenon. So according to VSEPR theory, the electron geometry of SF2 S F 2 molecule will be tetrahedral which includes all the valence electrons of the central atom as shown in the figure below. 120 degrees) with asymmetric charge distribution on the central atom.ġ09.5 degrees SF2 has a bond angle slightly less than 109.5 degrees due to its sp3 hybridization. SO2 - Sulfur Dioxide: The molecular geometry of SO2 is bent (approx.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
